
Key Takeaways The MLR review process is the compliance gatekeeper for every piece of pharma promotional content — and how efficiently teams run it directly determines time-to-market.
If your MLR process still runs on email chains and manual claims lookups, the bottleneck isn't your team — it's the infrastructure. |
|---|
Every piece of pharmaceutical promotional content, including sales aids, patient brochures, and digital ads, must pass through a structured approval workflow before it reaches a physician or patient. That workflow is the MLR review process: a cross-functional evaluation by Medical, Legal, and Regulatory teams that verifies scientific accuracy, legal defensibility, and regulatory compliance before any material goes to market.
The stakes are significant: in September 2025 alone, the FDA's Office of Prescription Drug Promotion issued more than 100 enforcement letters, both untitled letters and warning letters, to pharmaceutical companies for non-compliant promotional materials, signaling an enforcement posture that makes a rigorous promotional review process more critical than ever. The DOJ's False Claims Act enforcement data puts the stakes in sharp relief: since 1986, settlements and judgments have totaled more than $85 billion, with healthcare fraud, including pharmaceutical promotional violations, consistently the leading category.
For MLR operations leaders, promotional review managers, and the marketing and agency teams that support them, understanding how the process works — the stages, the standard roles, and where it most commonly breaks down — is the prerequisite for improving it. This guide covers all of that, including how AI is reshaping what's possible for teams managing growing content volumes with constrained reviewer capacity.
What Is MLR Review in Pharma?
MLR review — also called promotional review or promotional review committee (PRC) review — is the structured, multi-stakeholder process by which pharmaceutical and life sciences companies evaluate promotional materials before release.
The acronym reflects the three core functions involved: Medical, Legal, and Regulatory. Each brings a distinct lens to the same content, and all three must sign off before distribution. The scope is broader than many assume: in MLR review pharma settings, this covers everything from HCP sales aids to patient education pieces to digital ads — not just broadcast advertising.
It covers sales force materials, HCP-facing digital content, patient education pieces, disease awareness campaigns, conference presentations, social media posts, and website content — essentially any communication that makes a claim about a drug's efficacy, safety, or appropriate use.
Why MLR Review Exists
The financial and legal consequences of running a weak MLR review process in pharma are substantial and well-documented. GlaxoSmithKline's 2012 settlement of $3 billion, at the time the largest healthcare fraud settlement in US history, centered on off-label promotion and failure to report safety data to the FDA. Pfizer's 2009 settlement of $2.3 billion arose from similar promotional violations. A peer-reviewed analysis of FDA warning letters published in PMC found that omission of risk information was the most frequently cited violation in FDA enforcement letters, followed by unsubstantiated efficacy claims. These are the two categories that a well-run MLR review process is specifically designed to catch.
Beyond financial penalties, the reputational and operational impact of an untitled letter or warning letter can freeze entire promotional programs. In 2025, the FDA issued more than 100 enforcement letters in a single quarter, a historically unprecedented pace that signals a new era of scrutiny for pharma marketing operations.
How MLR Differs from Standard Content Review
Standard marketing review is primarily brand- and tone-focused. MLR review adds a compliance layer that requires medical, federal and international regulatory standards, and applicable legal precedents subject-matter expertise. Reviewers aren't checking style; they're assessing evidentiary support for each claim, verifying that safety disclosures are adequate, and confirming that the material won't create regulatory exposure.
This complexity is why the MLR process resists simple automation and why the organizations that run it most efficiently are those with tightly defined workflows, strong claims libraries, and clear role accountability at each stage.
What Does MLR Review Actually Evaluate?
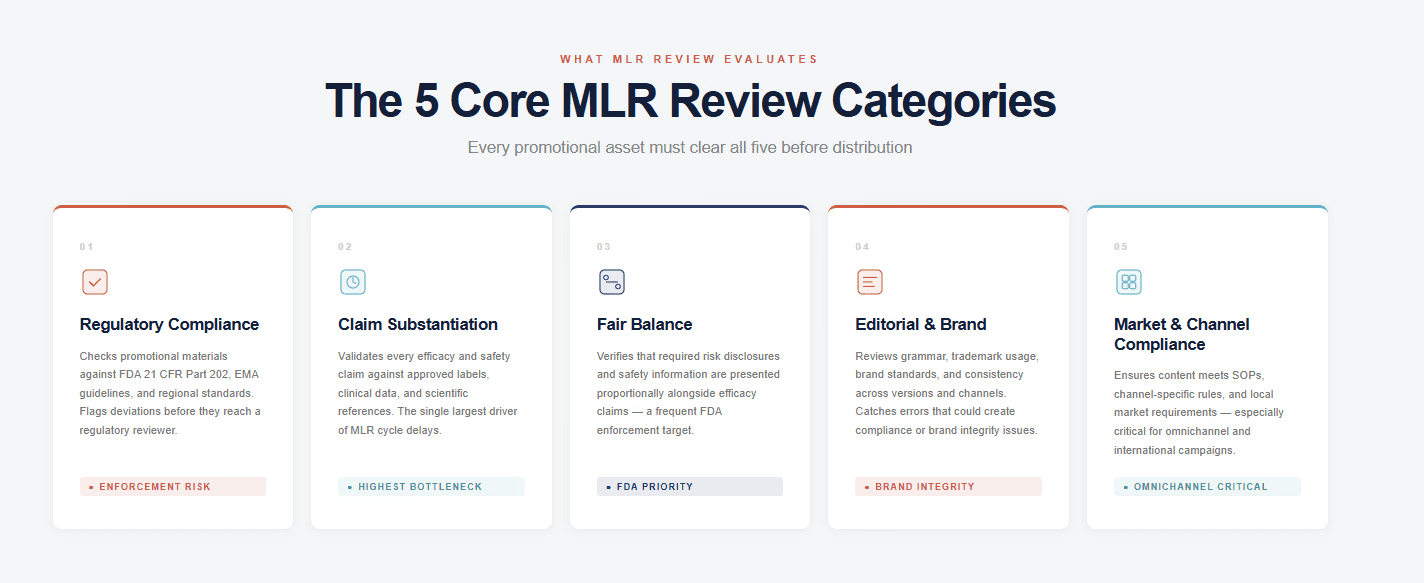
A comprehensive MLR review covers five distinct categories of compliance and accuracy. Many organizations address some of these well while leaving others underserved — which is why compliance gaps persist even in teams with mature review processes. Understanding all five matters: a single missed category can trigger re-review cycles or, worse, post-release enforcement. The five categories are:
Regulatory Compliance. Adherence to FDA, EMA, and applicable regional promotional standards. Includes assessment of off-label risk, required disclosures, and fair presentation of benefit/risk data.
Claim Substantiation. Verification that every efficacy or safety claim is supported by adequate, approved clinical evidence. This is consistently cited as the most time-intensive element of the MLR review process.
Fair Balance. Ensuring that safety information — including risks, warnings, and adverse events — is presented with appropriate prominence relative to benefit claims.
Editorial and Brand Guidelines. Confirmation of grammatical accuracy, trademark compliance, brand consistency, and adherence to internal style standards.
Market and Channel Compliance. Verification that the material meets channel-specific rules and local market requirements — particularly relevant for global campaigns and digital placements.
Understanding how specialized AI systems address each category differently is critical context for teams evaluating automation solutions.
Who Is Involved in the MLR Process?
The core review committee brings together three functions, each with a non-overlapping mandate. In practice, most organizations also include marketing and agency partners in an upstream role, preparing submissions and addressing feedback, even though they don't hold approval authority. Understanding what each reviewer is accountable for clarifies why sequential routing, while common, is one of the primary causes of cycle delays.
Reviewer | Primary Focus | Key Questions They're Answering |
|---|---|---|
Medical Reviewer | Scientific accuracy and clinical integrity | Are all claims evidence-based? Do they align with the approved label and clinical data? |
Legal Reviewer | Legal risk and liability exposure | Could this language create legal vulnerability? Does it comply with applicable laws? |
Regulatory Reviewer | FDA/EMA and market-specific compliance | Does this meet regulatory promotional standards? Are required disclosures present and adequate? |
Marketing / Agency | Brand alignment and content preparation | Is the content complete, on-brief, and submission-ready? Have prior comments been addressed? |
In large pharma organizations, each of these roles may involve multiple stakeholders and sub-reviewers, and a single piece of content commonly moves through numerous individual reviewers before final approval. This layered structure is why cycle times can stretch to weeks or months even for relatively simple materials.
What Are the Stages of the MLR Review Process?
While the exact steps vary across organizations, the promotional review workflow follows a recognizable sequence. Understanding these stages is essential for identifying where time is being lost and where workflow changes or AI assistance can have the greatest impact.
The following represents a standard workflow for a promotional asset moving from content development through final approval and submission.
Stage 1: Content Development and Internal Pre-Review
Marketing or agency teams develop the promotional material and conduct an internal editorial review before submission to the MLR committee. This stage should include verification that all claims are supported by approved references, that fair balance disclosures are present, and that the submission package is complete. Incomplete or low-quality submissions at this stage are the single most avoidable cause of MLR cycle delays. Every gap discovered during review sends the material back to this stage.
Stage 2: MLR Submission and Intake
The completed asset, along with its reference documents and annotation package, is formally submitted to the review queue. In organizations using a content management system like Veeva Vault PromoMats, this step is tracked digitally with timestamps, version control, and routing logic. In organizations relying on email-based workflows, this stage introduces the first major opportunity for version confusion and delayed acknowledgment.
Stage 3: Parallel or Sequential Review
Each reviewer, covering Medical, Legal, and Regulatory, evaluates the material against their specific criteria. Organizations differ on whether review is conducted sequentially or in parallel. Sequential review is more common but substantially slower; parallel review requires stronger coordination tools to prevent conflicting annotations and version collisions. Teams that have optimized their MLR workflows typically shift toward parallel review with structured reconciliation steps, reducing total cycle time without increasing compliance risk.
Stage 4: Reconciliation and Revision
Reviewers return comments, questions, and required changes to the content team. The material is revised and often re-submitted for a second round of review — particularly when substantive claims changes are required. Multiple revision cycles are normal; three or more rounds are common in complex therapeutic areas or for materials with high regulatory sensitivity. This loop is where the majority of overall cycle time is consumed.
Stage 5: Final Approval and Archival
Once all reviewers are satisfied, the material receives formal approval. In regulated markets, approved materials are archived with a complete audit trail covering reviewer identities, comment threads, revision history, and final sign-off timestamps. For US-market HCP materials, FDA Form 2253 submission requirements may also apply. This documentation is not administrative overhead; it is the compliance record that protects the organization in the event of a regulatory audit.
Why Does Speed Matter in the MLR Review Process?
Understanding who owns each stage of the MLR review process is only part of the picture. The other part is understanding what's actually at stake when that process runs slowly — because for pharma companies, delays in promotional review aren't just an operational inconvenience. They have direct commercial consequences.
In a market where competing brands are vying for the same physicians and patients, the timing of marketing materials matters. Companies that can move faster through the MLR review process gain meaningful advantages across several dimensions:
Establishing early brand recognition. Consistent, compliant messaging in market earlier gives a product more time to build awareness before competitors establish their own narrative.
Recouping development costs sooner. Drug development and production are expensive. Every week a product sits in review is a week of revenue not yet generated.
Securing a loyal consumer base. The earlier a company begins communicating with patients and HCPs, the longer it has to build trust and demonstrate value.
Differentiating from the competition. Being first to market with clear, substantiated messaging allows a brand to control the narrative in its therapeutic category before competitors do.
Capitalizing on emerging opportunities. Pharma markets shift — new data emerges, competitors stumble, patient needs evolve. Teams that can move through the MLR review process quickly are better positioned to respond.
Enhancing marketing performance. With an efficient MLR process in place, teams can make real-time adjustments to campaigns rather than waiting weeks for approval on updated materials.
None of this means cutting corners. It means that the MLR review process itself needs to be built for speed without sacrificing the accuracy and compliance it exists to protect. That tension is exactly where bottlenecks tend to surface.
What Are the Risks of Rushing the MLR Review Process?
Speed matters, but not at the expense of accuracy. The consequences of pushing non-compliant or inaccurate materials through the MLR review process range from manageable to severe, and they compound quickly.
The FDA's Office of Prescription Drug Promotion actively monitors promotional materials and issues enforcement actions when companies fall short. In September 2025 alone, the agency issued more than 100 untitled letters and warning letters to pharmaceutical companies, a signal that enforcement posture remains aggressive. Beyond regulatory action, the downstream risks of a rushed or inadequate MLR review include:
Legal exposure. Companies can face lawsuits for false advertising, copyright violations, or regulatory non-compliance. The DOJ's False Claims Act enforcement data shows healthcare fraud, including promotional violations, has generated more than $85 billion in settlements since 1986.
Compromised patient safety. A single inaccurate claim about dosing, contraindications, or side effects can put patients at risk. In a regulated industry, that's not a recoverable mistake.
Loss of scientific credibility. Once a company's standing within the medical and scientific community is damaged, rebuilding trust is a long process — often extending well beyond the product at issue.
Increased operational costs. Errors caught after release divert resources from strategic initiatives into rework, retraining, and remediation. The cost of remediation routinely exceeds what proper upfront review would have required.
The goal, then, isn't to choose between speed and accuracy in the MLR review process. It's to build a process — and invest in the right infrastructure — that doesn't force that tradeoff in the first place.
5 Common Bottlenecks in the MLR Review Process
These are the friction points that consistently extend cycle times across mid-to-large pharma organizations and agencies. They're well-known problems — and they're also solvable with the right combination of process discipline and technology.
Claims substantiation delays. Locating, reviewing, and annotating clinical references for each claim is manually intensive and often relies on reviewers searching fragmented document libraries. When references are hard to find or out of date, review stalls. A dynamic, searchable claims library addresses this directly, and building one is increasingly feasible with AI-assisted claims extraction.
Incomplete submissions entering the queue. Materials submitted without complete reference packages, missing safety disclosures, or unresolved prior-round comments create immediate re-routing. Each returned submission adds days to the cycle and consumes reviewer capacity on content that wasn't ready.
Sequential review routing. When Medical, Legal, and Regulatory reviewers must complete their reviews in sequence rather than in parallel, total cycle time is the sum of each reviewer's turnaround. For complex materials, this can add weeks. Shifting to parallel review with conflict-resolution workflows is one of the highest-impact structural changes available.
Version control failures. Email-based workflows routinely produce multiple simultaneous draft versions with contradictory comments. Reconciling these versions requires manual effort and introduces the risk of applying the wrong feedback — or missing required changes entirely. Centralized content management systems eliminate this problem at the infrastructure level.
Growing content volume outpacing reviewer capacity. Digital marketing programs, omnichannel campaigns, and personalized HCP engagement have dramatically increased the volume of materials requiring MLR approval. Most teams' reviewer headcount has not scaled proportionally. This capacity gap is the structural driver behind most cycle time deterioration in recent years, and it's the problem AI-assisted review is best positioned to address.
How Is AI Changing the MLR Review Process?
The application of AI to the MLR process is moving beyond pilot programs and into operational deployments. The most credible use cases aren't about replacing reviewers. They're about handling the volume and consistency work that currently delays qualified reviewers from doing the high-judgment work only they can do.
The most impactful applications include automated pre-screening for completeness and obvious compliance gaps before content enters the formal queue, AI-assisted claims substantiation that matches draft claims against approved clinical references and flags gaps, and continuous fair balance checking that tracks disclosure adequacy across content updates.
Human-in-the-Loop Remains Non-Negotiable
The distinction between AI augmentation and AI replacement matters for both regulatory and operational reasons. AI systems excel at pattern recognition, consistency checking, and high-volume scanning. As noted in Responsible AI in Healthcare, the most effective implementations keep qualified reviewers accountable for all final approvals, with AI surfacing issues, not deciding them.
Teams that have optimized MLR workflows with AI and parallel routing report substantial cycle time reductions. Industry analysis from EY Aqurance found that companies with optimized workflows achieved a 57% reduction in review cycle times. For teams deploying comprehensive AI-assisted review across all five MLR categories, gains can be substantially higher.
Approach | Typical Cycle Time |
|---|---|
Manual / email-based MLR | 6–12 weeks |
CMS-based MLR (e.g., Veeva Vault) | 3–6 weeks |
AI-assisted MLR with human oversight | Days to 1–2 weeks |
For teams evaluating AI options, the key questions aren't about speed alone. Coverage matters: specifically, whether the AI solution addresses all five MLR review categories or only a subset. Solutions that address regulatory and claims review but leave fair balance, editorial, and channel compliance to manual passes still leave significant cycle time on the table. Understanding the difference between general and specialized AI is essential when assessing which tools will produce durable gains versus surface-level improvements.
FAQ
What is MLR review in pharma, and what does it stand for?
MLR stands for Medical, Legal, and Regulatory. In pharma, MLR review is the structured evaluation process that every piece of promotional content must pass through before it can be distributed to healthcare professionals, patients, or the public. It ensures claims are scientifically substantiated, legally defensible, and compliant with FDA and EMA promotional standards. The process is also commonly referred to as promotional review or PRC (Promotional Review Committee) review.
In organizations relying on manual or email-based workflows, review cycles typically run six to twelve weeks for complex materials. Teams with structured content management systems and optimized routing often reduce this to three to six weeks. Organizations that have implemented AI-assisted review with parallel routing report cycles as short as a few days for lower-complexity content. The variance is primarily a function of workflow design, submission quality, and reviewer capacity — not the complexity of the content itself.
What is the difference between MLR review and regulatory submission?
MLR review is an internal process — it happens before a material is used or distributed. Regulatory submission (such as FDA Form 2253 filing for US HCP materials) happens after MLR approval and is a formal notification to the regulator that a promotional piece has been released. Passing internal MLR review does not automatically satisfy external submission requirements; both must occur for a material to be fully compliant in regulated markets.
What are the most common reasons MLR review cycles take longer than expected?
The most consistent drivers of extended cycles are incomplete submissions entering the review queue, sequential rather than parallel reviewer routing, absence of a current claims library requiring manual reference searches, and growing content volume that outpaces available reviewer capacity. Organizations that systematically address all four of these factors — through submission standards, routing redesign, claims library investment, and AI-assisted volume handling — consistently achieve the greatest reductions in cycle time.

The MLR Process Hasn't Changed — But the Cost of Running It Slowly Has
What MLR review requires hasn't fundamentally shifted in decades: Medical, Legal, and Regulatory teams evaluating promotional content against a defined compliance standard before release.
What has changed is the volume of content requiring review, the speed at which markets move, and the organizational cost of every day a reviewed-and-approved piece isn't in market. Review cycles that were once an accepted cost of doing business are now measurable competitive disadvantages, tracked in missed HCP engagement windows, delayed launches, and brand teams spending more time in feedback loops than in market.
Teams that are closing this gap share a common approach: structured workflows, strong claims infrastructure, and AI-assisted review that amplifies reviewer capacity without removing reviewer accountability. Revisto is built specifically for this problem — an AI-powered platform that addresses all five MLR review categories and deploys within your existing workflow, including directly inside Veeva Vault PromoMats, so there's no learning curve and no disruption to how your team already works. Request a demo to see how AI-assisted review can reduce your MLR cycle times without compromising the human judgment that compliance requires.